Filament Free 3D Printing Microfactory for Oral Solid Dosage Forms
Introduction
About 80% of all drugs are orally administered with tablets or oral solid dosage forms (OSD) the preferred product presentation. There are multiple reasons for this situation covering patient acceptability, self administration, ease of distribution and product stability. However, OSD manufacture from a dry powder mixture by physical compression between two punches constrained by a die has not changed since its introduction almost 200 years ago.
OSD manufacture requires the mixing of the powdered drug (active pharmaceutical ingredient, API) with usually multiple excipients (diluent, binder & disintegrant), followed by processing to produce an intermediate granular stage. The granules require mixing with further excipients (disintegrant, glidant & lubricant) before the final compression stage. The complete process takes weeks and any formulation changes, for example a change in API dose or the excipients requires re-manufacture. Manufacture is therefore time consuming and in situations where it sits on a critical project path induces delays.
Additive Manufacturing (AM)/3D Printing (3DP)
The advent of computers, computer aided design and specialist printers, has led through the 1970’s and 80’s to the introduction of 3D printing (3DP) or additive manufacturing (AM) techniques. Various approaches can be applied and in the initial stages AM systems were utilised for rapid prototyping or for the rapid on demand production of one off articles. The principle is based on the creation of a 3D object using computer aided design (CAD), the computer can then horizontally slice and digitise the object into layers, which represent the overall structure. The digital layers can then be sequentially used to drive a printer which lays down the layers on top of each other to re-create the CAD object in physical form. This allows the precise and repeatable creation of objects with complex structures and current 3D printing processes are considered viable as an industrial-production process. Fused deposition modelling (FDM™), which uses a continuous thermoplastic polymer filament is the most common 3D printing process. The potential for application to the manufacture of OSD is obvious but to date only one marketed pharmaceutical OSD using a powder bed printing process is available.
Fused Deposition Modelling
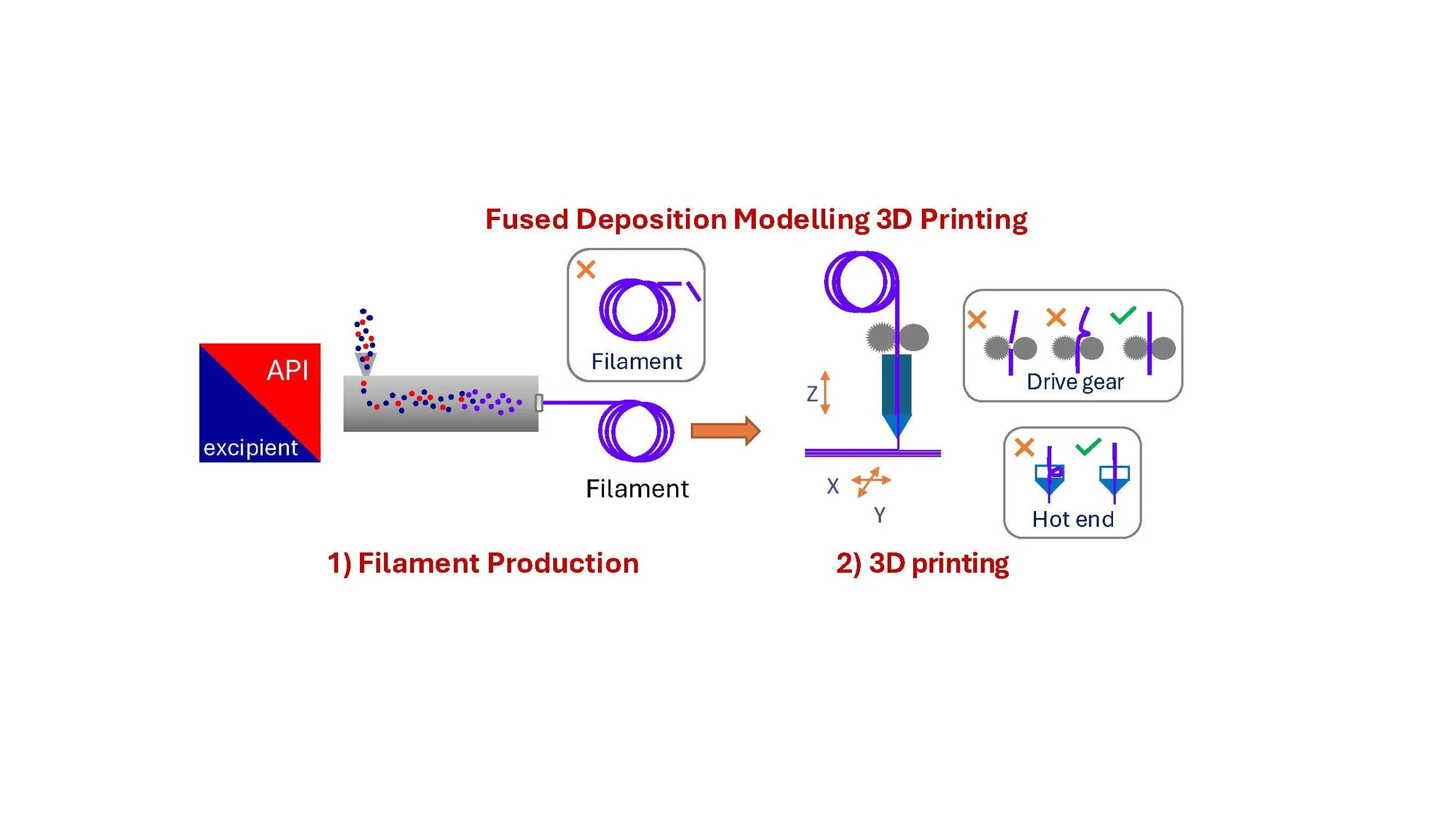
Fused Deposition Modelling also known as Fused Filament Fabrication (FFF) is one of the most common AM or 3DP techniques and multiple research groups have applied this to the pharmaceutical production of OSD. The process requires two stages (Figure 1) the initial manufacture of a polymer filament, which the printer then utilises as an “ink” to deposit on a print bed. The OSD formulation therefore must be suitable for both processes. Filament formation is usually conducted using a heated extrusion device which melts the polymer and drug before extruding through a circular die to create the filament. The filament ink is then fed by mechanical drive wheels into a hot end printing head where it re-melts prior to deposition on the print bed.
Figure 1. Schematic of Fused Filament Fabrication 3D Printing Process
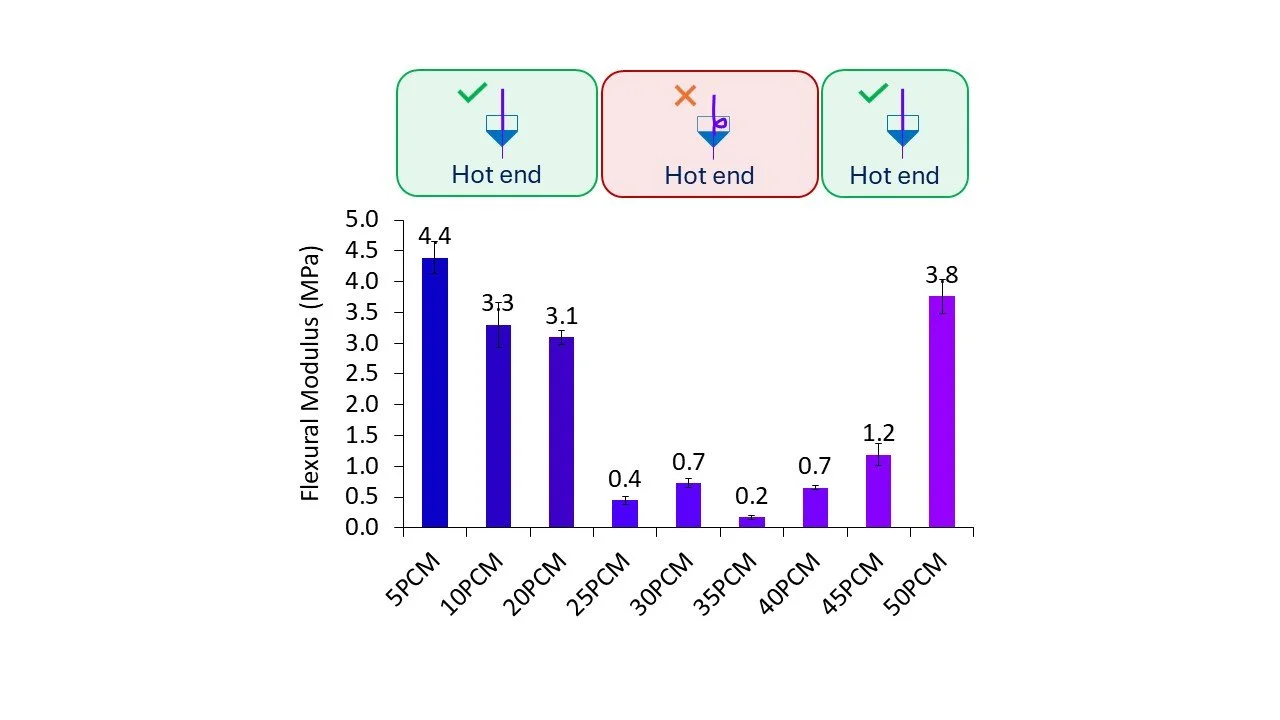
Overall this system has multiple failure points (Figure 1) where the formulation’s physicochemical properties either in the solid or liquid state are not suitable (Figure 2). This requires a two stage formulation development process (filament then printing), restricts the formulation space due to process requirements, and influences the final OSD property’s e.g. potential dose content and dissolution performance.
Figure 2. Filament Failure – Paracetamol/Affinisol (see Prasad, Additive Manufacturing 29 (2019) 100776.
Filament Free 3DP
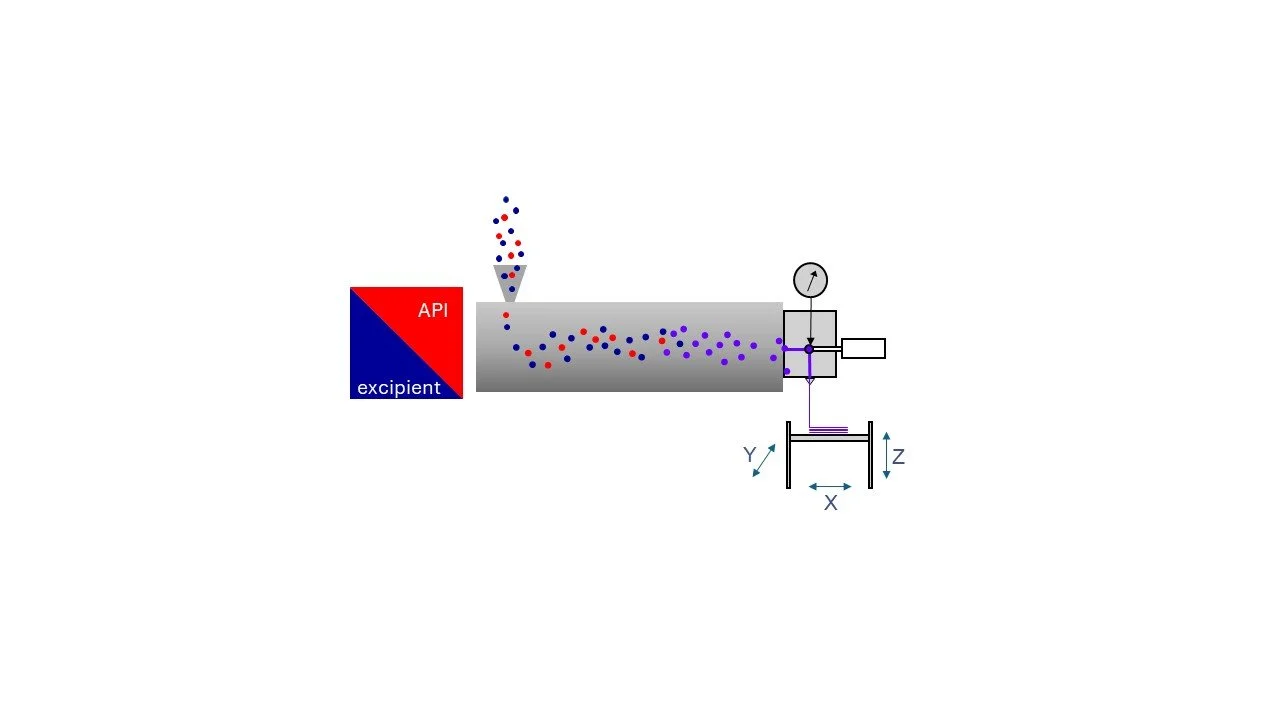
In order to remove the limitations of FDM 3DP Adaptim3D developed a filament free microfactory (Figure 3) capable of direct printing from the extruder using a patented print head (see Prasad Additive Manufacturing 77 (2023) 103803). This advance accelerates the process permitting a focus on the OSD’s formulation and final properties e.g. dissolution. The microfactory is a bench top device and also reduces the manufacturing footprint and resources required. Control of the OSD’s microstructure via 3DP permits the selection of a desired dissolution profile. Or the polymer matrix can be selected to control dissolution.
Figure 3. AdaptiM3D Filament Free 3DP
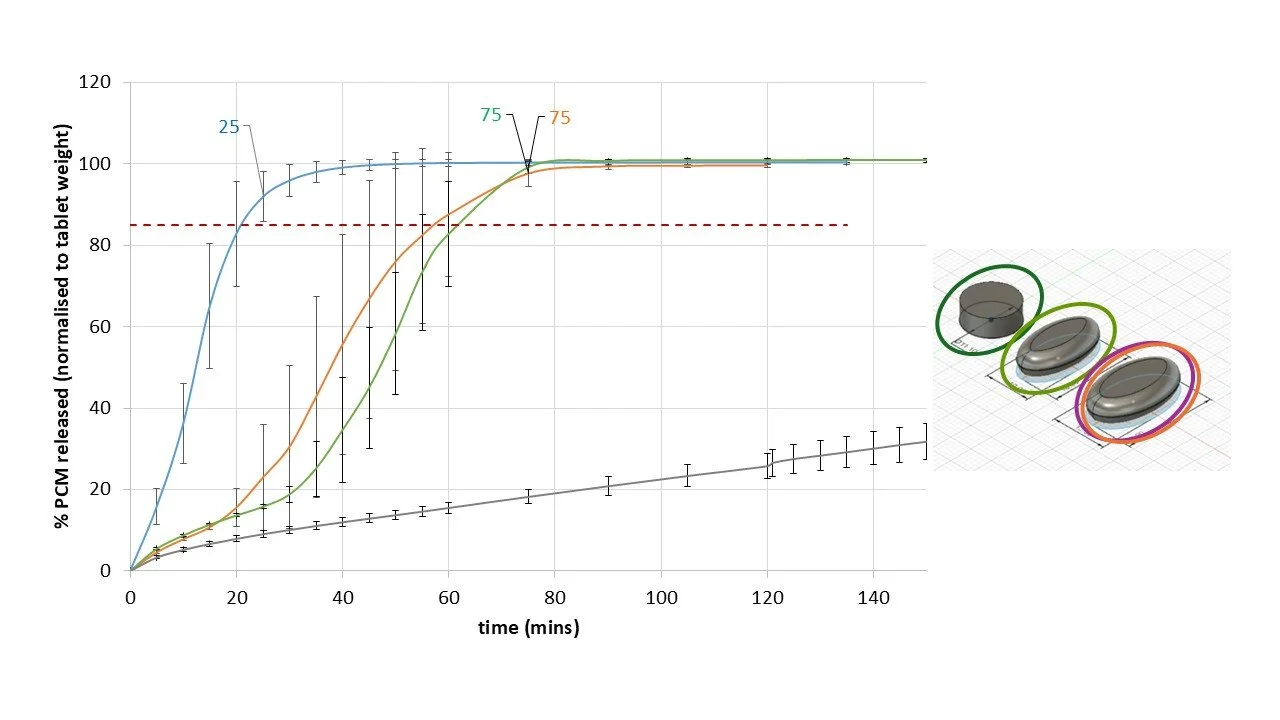
Figure 4 presents the dissolution profiles of a 1:1 w/w paracetamol (PCM)/Affinisol (hydroxypropylmethylcellulose) formulation printed using the filament free microfactory. Dissolution performance control, ranging from rapid to sustained, within a single formulation can be simply controlled by variation of OSD microstructure and shape.
Figure 4. Dissolution of various paracetamol OSD controlled via 3DP microstructure.
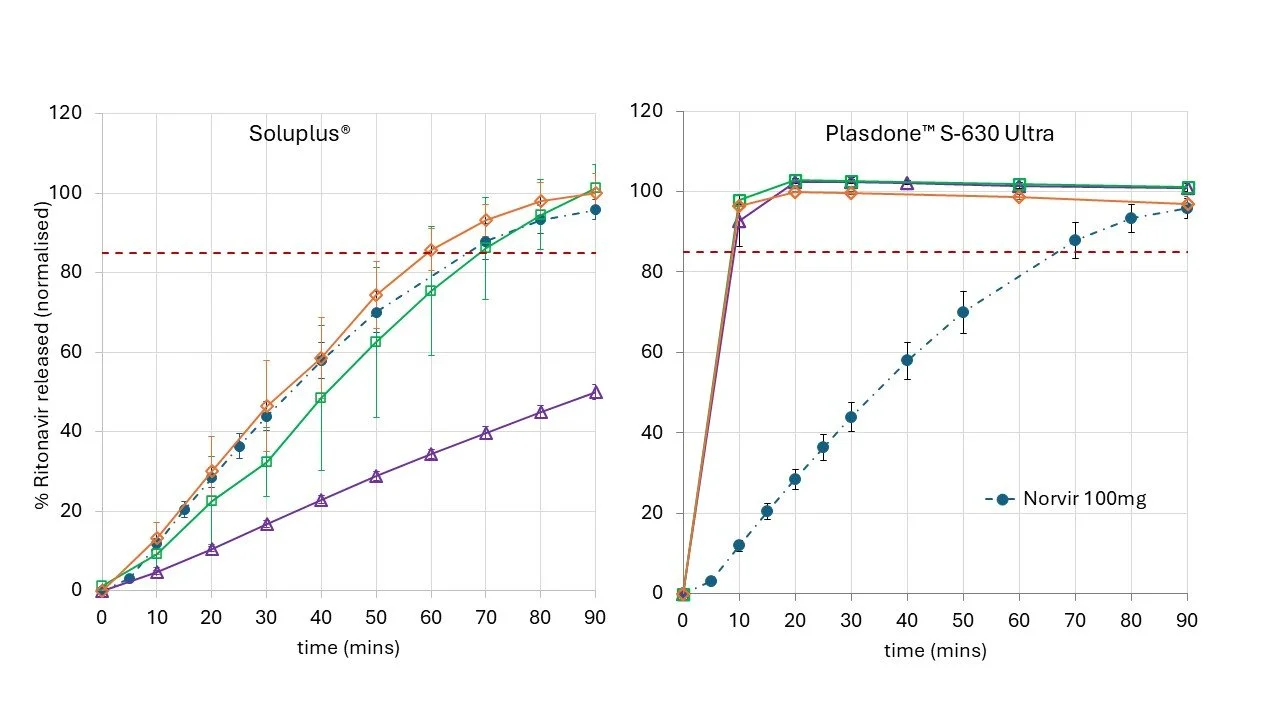
Figure 5 presents the dissolution profiles of two ritonavir OSD formulations compared against the marketed product (Norvir™). Judicious choice of polymer excipient and infill allows preparation of OSD systems that can match, improve or control drug release when compared with the marketed product.
Figure 5. Dissolution of two ritonavir OSD formulation vs marketed product (Norvir).
Summary
The AdaptiM3D 3DP microfactory opens up the pharmaceutical formulation space and facilitates a single, streamlined 3DP process with reduced thermal load. This allows rapid formulation, design of custom dissolution profiles and establishment of a formulation/OSD design space suitable for agile manufacture.
Next in this technical series: Simple assessment of polymer drug mix rheology rapidly identifies processing space.